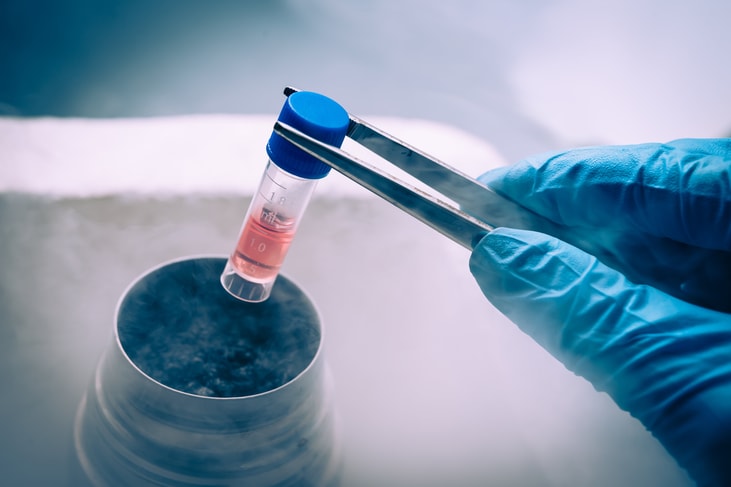
Marken adds cryogenic storage services
Supply chain provider Marken today said it will double the space of its GMP facility in Frankfurt to further expand its frozen room storage capabilities in anticipation of the Covid-19 vaccine and storage needs.
In addition, Marken announced the integration of its cryogenic storage services which went live in June in both Frankfurt and Philadelphia.
These latest additions to the Marken GMP network service portfolio further reinforces the company’s dedication to serving its clients as a complete, end-to-end service provider. Future cryogenic storage locations will include the APAC region.
Offering a comprehensive cryogenic transport and storage solution enables cell and gene therapy companies to simplify their supply chain by allowing them to consolidate processes with Marken, an established and trusted service provider. Fewer handoffs mean fewer variables, less risk and an unbroken chain of custody.
... to continue reading you must be subscribed