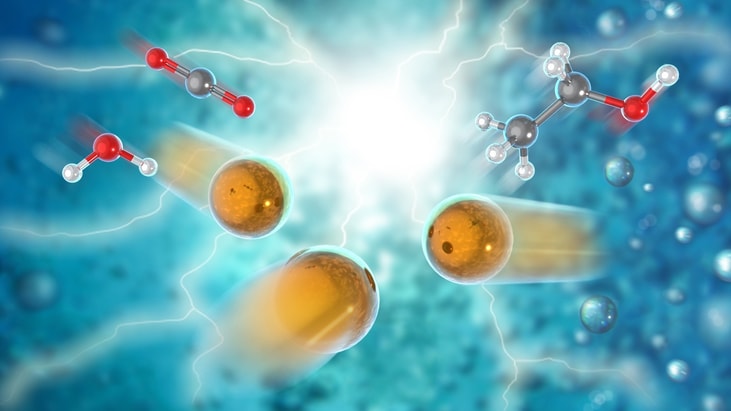
Turning carbon dioxide into liquid fuel
US researchers have discovered a new electrocatalyst that converts carbon dioxide (CO2) and water into ethanol with very high energy efficiency, high selectivity for the desired final product and low cost.
Led by the US Department of Energy’s (DOE) Argonne National Laboratory in collaboration with Northern Illinois University, the research teams process electrochemically converts the carbon dioxide emitted from industrial processes, such as fossil fuel power plants or alcohol fermentation plants, into valuable commodities at reasonable cost.
Ethanol is a particularly desirable commodity because it is an ingredient in nearly all US gasoline and is widely used as an intermediate product in the chemical, pharmaceutical and cosmetics industries.
... to continue reading you must be subscribed